 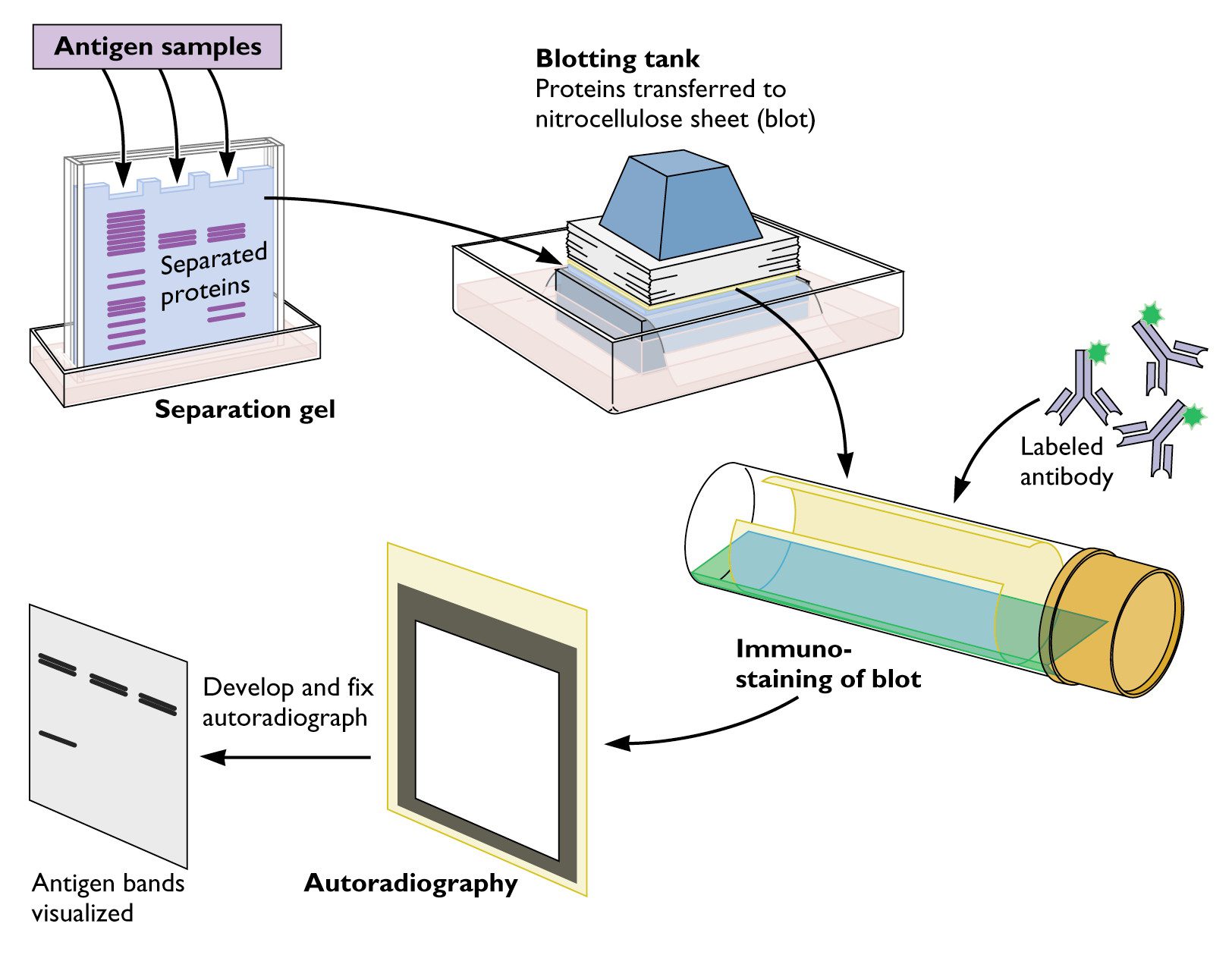
A synthetic peptide is added to detect antibodies to the HIV-1 group O. The ADVIA Centaur CHIV assay uses recombinant antigens, including an HIV-1 envelope protein (gp41/120) and an HIV-2 envelope protein (gp36), and three monoclonal antibodies specific to HIV p24 antigen. The ADVIA Centaur CHIV assay is an antigen-bridging, magnetic microparticle-based, chemiluminometric immunoassay that detects antibodies against HIV-1 group M and O, HIV-2, and p24 antigens in the serum or plasma. Serum samples from 100 hospitalized and 600 healthy subjects were included during March 2012. The 54 HIV false-positive and 14 known HIV-positive serum samples were collected from March 2010 to March 2012. These known HIV-positive samples yielded both a gp160/gp120/gp41 band and at least one p24 or p31 band on western blot analysis. 1) and two Korea Food & Drug Administration (KFDA) standard panels (09/030 8 and 16) containing eight HIV-positive, ten indeterminate, and four negative serum samples. The accuracy of the CHIV assay was evaluated using 14 known HIV-positive serum samples, which repeatedly produced reactive results on EHIV assay and were confirmed HIV positive by western blot analysis ( Fig. All samples from 100 hospitalized and 600 healthy subjects were classified as HIV-negative, with nonreactive results on EHIV assay. Serum samples from 100 consecutive patients hospitalized in the Department of Internal Medicine and the intensive care unit of the Samsung Medical Center and 600 consecutive healthy subjects who visited the health promotion center for routine checkup were included to determine specificity. To evaluate the false-positivity and specificity of the CHIV assay, two sets of samples were tested: a batch of HIV false-positive and HIV-negative serum samples from hospitalized patients and healthy subjects who requested an HIV antibody test, and 54 HIV false-positive serum samples, which repeatedly tested reactive on EHIV assay, but produced negative results on western blot analysis ( Fig. Newer fourth-generation HIV assays, however, have improved on the limitations of previous assays, while reducing the seroconversion window. The specificity of fourth-generation assays was reported to be lower than that of third-generation assays for screening blood donors, and the high rate of false-positive results (0.2%) resulted in difficulties in blood donor screening. The risk of interference was also predicted to be higher, because both serological markers were determined in one test well. Early fourth-generation assays had limited ability to detect HIV antigen compared to single p24 antigen assays. Fourth-generation assays have been reported to possess potential non-specific reactivity, because two principles are combined in one assay. A negative result for p24 antigen assay after antibody seroconversion does not exclude the possibility of HIV infection.Ĭombining both antigen and antibody detection increases sensitivity in the early seroconversion window, while improving the chances of identification of low-titer anti-HIV antibodies and persistent antigenemia in the late stage. However, the p24 antigen titer continuously decreases after reaching its peak and is usually undetectable. The HIV p24 antigen assay can detect p24 antigen, beginning approximately on the 15th day and peaking between 20 to 30 days, thus enabling early diagnosis of HIV infection. The window period, from the presence of HIV-1 RNA in plasma to antibody seroconversion, varies between 10 and 27 days, depending on the route of infection. When patients are first exposed to HIV, serum HIV antibodies are produced these can be detected at least 3 weeks later by third-generation antibody assays. Recently published guidelines include a routine HIV screening test, which can benefit the patients by detecting HIV before seroconversion. Early diagnosis of HIV infection reduces morbidity and mortality and minimizes potential transmission of HIV. A recent study showed that delayed diagnosis of HIV and late presentation were major risk factors of early death in Korea, and 41% of the newly diagnosed HIV-infected individuals presented with advanced HIV disease. The rate of HIV infection is constantly increasing, with 888 new infections being reported in 2011. In 2008, HIV was estimated to cause 2.7 million new infections and 2 million deaths worldwide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
